Printed bakery bags or coloured napkins: No health impairments are expected from the release of certain colourant components New data allow for an updated assessment of naphthol AS, NAAX, NDPA and HNS in food contact materials
Replaces Opinion No. 037/2019 of 26 September 2019 (in German only)
What it's about:
Materials that come into contact with food as intended or as can be expected are also known as food contact materials or food contact articles. In addition to cardboard packaging, bakery bags and napkins, food contact materials made of paper and cardboard also include other utensils such as muffin tins and drinking straws. Numerous additives and finishing agents are used in their production. One example is colourants, which are used to dye or (colour) print on some of these products.
In this risk assessment, the German Federal Institute for Risk Assessment (BfRshort forGerman Federal Institute for Risk Assessment) has examined, on the basis of new data on stability and toxicity, whether the intake of certain substances in colourants (naphthol AS, HNS, NAAX and NDPA) poses health risks to consumers. According to the assessment, this is not to be expected for the detected amounts of these substances.
Naphthol AS, NAAX, NDPA and HNS are very likely to be starting materials, impurities or degradation products of certain colourants, known as azo dyes and pigments. To determine the amounts of these substances that may migrate from food contact materials into food, the researchers used the cold water extraction method. The samples were placed in contact with water – representing food – at room temperature for 24 hours. The concentrations of the four substances in the water were then determined.
There are no legally defined migration limits or derived health-based guidance values for any of the four compounds. The BfRshort forGerman Federal Institute for Risk Assessment therefore derived acceptable intake values based on toxicological data on the compounds mentioned and on substances with a similar chemical structure, using computer-aided prediction models.
According to these values, no adverse health impairments are to be expected for a 60 kgshort forkilogram person with a lifetime intake of 360 µgshort formicrogram (micrograms) per day of HNS. The amounts of HNS migrating from the samples examined do not exceed this value and therefore do not pose an increased health risk.
For the substances naphthol AS, NAAX and NDPA, the data available to date has not been sufficient for a health risk assessment. As stated in External Link:earlier version of this opinion (in German only, No. 37/2019), there were also indications that these substances or their possible metabolites might have mutagenic and carcinogenic properties in the human organism. The new data on possible metabolites that are now available show that the breakdown of these substances into mutagenic metabolites occurs only to a very limited extent, if at all. The previously unavailable and now newly available tests on NDPA showed no mutagenic effects. According to current data, migration of naphthol AS, NAAX and NDPA of up to 50 micrograms (µgshort formicrogram) per kilogram (kgshort forkilogram) of food does not pose any health risks to consumers. Only in individual samples (2 out of a total of 51 samples tested) a migration above 50 µgshort formicrogram/kgshort forkilogram of food was observed for naphthol AS. The toxicological data currently available is insufficient to perform a health risk assessment for this migration. The BfRshort forGerman Federal Institute for Risk Assessment therefore recommends that no migration of naphthol AS (or NAAX and NDPA) above 50 µgshort formicrogram/kgshort forkilogram food should occur.
Packaging paper for baked goods often contained several of the substances examined and released them into the water extract. Other sample groups, such as drinking straws, muffin tins and napkins, showed significantly fewer and significantly lower releases overall. In each sample group, there were also samples in which no detection of naphthol AS, NAAX and NDPA was possible. The BfRshort forGerman Federal Institute for Risk Assessment recommends that manufacturers test their raw materials and end products for impurities containing these substances or their release and, if necessary, seek alternatives to the materials used.
1 Subject of the assessment
In the following, the BfRshort forGerman Federal Institute for Risk Assessment assesses the available food monitoring data from 2018 on the release of the compounds naphthol AS (CAS 92-77-3, also 3-hydroxy-2-naphthanilide), N-acetoacetyl-m-xylidine (CAS 97-36-9, NAAX), N-(2,4-dimethylphenyl)-acetamide (CAS 2050-43-3, NDPA) and 3-hydroxy-2-naphthoic acid (CAS 92-70-6, HNS) from various food contact materials (FCM) made of paper and cardboard in cold water extracts. The regional authority of a German federal state examined the cold water extracts of various FCM made of paper and cardboard, such as napkins, cardboard packaging and drinking straws, for residues of dyes, their degradation products and manufacturing-related impurities. The cold water extract was prepared in accordance with DIN EN 645 and analysed using LC-MS/MS. The four compounds mentioned (Figure 1) were quantitatively determined in some samples.
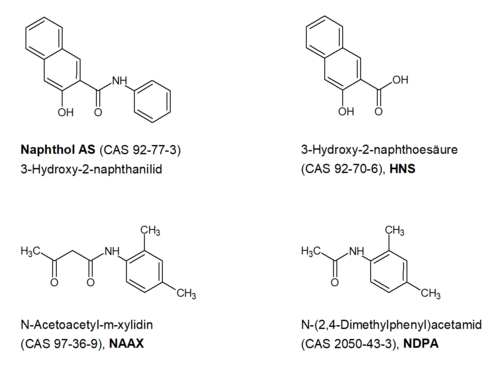
The BfRshort forGerman Federal Institute for Risk Assessment had already carried out a health risk assessment of the amounts of these substances determined in the migration experiments in 2019. Based on newly submitted data on the stability and toxicity of some of the compounds, the BfRshort forGerman Federal Institute for Risk Assessment has reassessed the release amounts.
2 Result
There are no legal or normative limit values for the release of the compounds shown in Figure 1 from paper and cardboard. The BfRshort forGerman Federal Institute for Risk Assessment has derived acceptable intake levels based on toxicological data on the compounds mentioned and chemically/structurally similar compounds, as well as in silico data.
According to these values, no impairments are to be expected from an intake of 360 µgshort formicrogram HNS per day for a 60 kgshort forkilogram person.
For naphthol AS, NAAX and NDPA, the BfRshort forGerman Federal Institute for Risk Assessment has derived from the available data that any release of presumably genotoxic or carcinogenic aniline derivatives, if any, occurs at amounts that do not give rise to any general cause for increased health concern: As there are no comprehensive long-term in vivo toxicological studies on naphthol AS, NAAX and NDPA, the assessment was based on an in vitro study and the "threshold of toxicological concern" (TTCshort forThreshold of Toxicological Concern) concept. According to the threshold of toxicological concern (TTCshort forThreshold of Toxicological Concern) concept, daily intake of a genotoxic carcinogenic substance of no more than 0.15 µgshort formicrogram per person does not give rise to increased concern. For non-genotoxic substances in the FCM domain, a daily intake of up to 50 µgshort formicrogram per person is acceptable in terms of health according to the "Note for Guidance" of the European Food Safety Authority (EFSAshort forEuropean Food Safety Authority) (EFSAshort forEuropean Food Safety Authority Panel on Food Additives et al.short foret alii (lat. "and others") 2008) . Based on the typical assumption (consumption of one kilogram of food that has been in contact with FCM per day), this results in a tolerable migration of up to 50 µgshort formicrogram per kgshort forkilogram of food.
HNS, NAAX and NDPA were detectable in almost half of the 51 samples examined, while naphthol AS was found in 10 samples. Particularly striking was packaging paper for baked goods, which often contained several of the substances and released them into the water extract. Other sample groups, such as drinking straws, muffin tins and napkins, showed significantly fewer and significantly lower releases overall. In each sample group, there were samples in which naphthol AS, NAAX and NDPA were not detected. The measured migration values for HNS, NAAX and NDPA were all less than 360 µgshort formicrogram/l and less than 50 µgshort formicrogram/l, respectively. Accordingly, these migration values do not pose an increased health risk. For individual samples (n = 2), migration above 50 µgshort formicrogram/kgshort forkilogram food was observed for naphthol AS. The toxicological data currently available is insufficient to perform a health risk assessment for this migration. The BfRshort forGerman Federal Institute for Risk Assessment therefore recommends that no migration of naphthol AS (or NAAX and NDPA) above 50 µgshort formicrogram/kgshort forkilogram food should occur. Manufacturers should test their raw materials and end products for impurities from these substances or their release and, if necessary, seek alternatives to the materials used.
3 Rationale
3.1 Risk assessment
3.1.1 Hazard identification
3.1.1.1 Potential hazards
In the production of paper and cardboard, which are to be further processed into various food contact materials, a variety of auxiliary and finishing agents are used to produce the properties desired for the respective application. The four substances naphthol AS, HNS, NAAX and NDPA have a very high probability of belonging to the group of starting materials, impurities or degradation products of (azo) dyes. This is indicated by the listing of NAAX and 2-naphthol in Annex 10 on printing inks of the Swiss Commodities Ordinance (EDI 2017) and by the chemical structure of the four compounds.
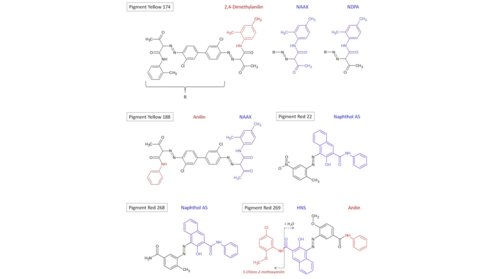
For example, the pigments Yellow 174 (CAS 78952-72-4), Yellow 188 (CAS 23792-68-9), Red 22 (CAS 6448-95-9), Red 268 (CAS 16403-84-2) and Red 269 (CAS 67990-05-0) contain one or more of the four compounds mentioned above as substructures (see Figure 2). The pigments mentioned are also listed in Annex 10 of the Swiss Commodities Ordinance (EDI, 2017) and, in part, in Annex 14 of the German Commodities Ordinance.
In addition to naphthol AS, NAAX, NDPA and HNS, the compounds aniline (CAS 62-53-3), 2,4-dimethylaniline (CAS 95-68-1) and 5-chloro-2-methoxyaniline (CAS 95-03-4) were also determined in the cold water extracts of some of the samples examined, which are also expected degradation products or impurities of some of the pigments shown in Figure 2 or of naphthol AS, NAAX and NDPA. The aforementioned degradation products or impurities of dyes or pigments may transfer to food upon contact and be taken up by consumers in an oral manner.
3.1.2 Hazard characterisation
3.1.2.1 Intake, distribution, metabolism, excretion
The BfRshort forGerman Federal Institute for Risk Assessment has no data on toxicokinetics for any of the four compounds. The "rule of five" (Lipinski et al.short foret alii (lat. "and others") 2001) can be used to theoretically estimate the possible absorption of the compounds. According to this rule, readily absorbable substances have a molar mass of less than 500 g/mol and no more than five hydrogen bond donors, no more than ten hydrogen bond acceptors and an octanol/water partition coefficient (LogPOW ) of approximately between 0 and 5. This applies to all four compounds considered (see Table 1 ).
Table 1
Structural and physicochemical data for estimating oral absorption according to the "rule of five"
| Naphthol AS | HNS | NAAX | NDPA | |
| Molar mass in g/mol | 263,3 | 188,2 | 205,3 | 163,2 |
| Number of hydrogen bond donors | 2 | 2 | 1 | 1 |
| Number of hydrogen bond acceptors | 1 | 1 | 2 | 1 |
| LogPOW* | 3,36 | 2,11 | 2,06 | 2,03 |
*Calculated using: External Link:http://www.swissadme.ch/index.php
Since systemic toxicity has also been reported in the literature for each of the substances or for chemically/structurally similar compounds in various studies (see below), it can be assumed that all four compounds are absorbed into the blood via the gastrointestinal tract in relevant amounts. For naphthol AS, NAAX and NDPA, the question of metabolism and the associated cleavage of the amide bond is of great importance, as this could lead to the formation of the primary aromatic amines aniline and 2,4-dimethylaniline. Aniline is classified as a class 2 mutagen and carcinogen according to Regulation (EC) No 1272/2008. 2,4-Dimethylaniline is also suspected of being mutagenic and carcinogenic (BAuA 2016) (see below).
Naphthol AS: According to the in silico prediction of metabolism using the OECD Toolbox Version 4.6, there is no occurrence of cleavage of the amide bond. However, the fact that the cleavage product HNS was found in a large number of samples and that the cleavage product aniline was also detected in one sample (see sections 2 and 3.1.1) could be due to the fact that the compound is possibly unstable enough to be cleaved during or after technical application. The possible cleavage of naphthol AS into the primary aromatic amine aniline after oral intake during digestion was investigated in two different in vitro models (Bittner et al.short foret alii (lat. "and others") 2022) . One model followed the recommendations of the European Food Safety Authority (EFSAshort forEuropean Food Safety Authority), while the other model was developed by the Dutch National Institute for Public Health and the Environment (RIVM). The protocol recommended by EFSAshort forEuropean Food Safety Authority involves stricter conditions and a longer incubation period. The two models simulate the successive effects of saliva, gastric juice and intestinal fluid on the substances. After incubation of naphthol AS in both digestion models, possible degradation products were analysed using LC-MS/MS. Before the experiment began, a concentration of 0.09% aniline was measured as an impurity in naphthol AS. Beyond this background level, no increase in aniline concentration was observed in either the EFSAshort forEuropean Food Safety Authority model or the RIVM model. It can therefore be concluded that under the selected conditions, no detectable hydrolysis of naphthol AS to aniline takes place.
NAAX: In vivo, after oral administration of NAAX to rats, the formation of 2,4-dimethylaniline and its binding to haemoglobin molecules in the rats' blood was observed (BUA 1992; Sabbioni 1999). However, the study does not authorise any conclusions to be drawn about the metabolic rate. In silico data generated using the OECD Toolbox Version 4.6 support the in vivo findings, as 2,4-dimethylaniline is predicted to be a possible metabolite. A study by Rasper (2016) also showed that NAAX is cleaved at 80 °Cshort fordegrees Celsius in an aqueous solution – without enzymatic action – and that the lower the pH value of the solution, the greater the cleavage. The 2,4-dimethylaniline that forms during this cleavage was detected by LUA Saxony in two samples in addition to NAAX (see sections 2 and 3.1.1).
In addition, a recent study used in vitro digestion models to investigate the possible cleavage of NAAX to 2,4-DMA (Bittner et al.short foret alii (lat. "and others") 2022) . For this purpose, two different in vitro digestion models were used, as described above, one from EFSAshort forEuropean Food Safety Authority and the other from RIVM. The EFSAshort forEuropean Food Safety Authority digestion model showed that NAAX was cleaved to 2,4-DMA in both saliva and gastric juice, but not in intestinal fluid. The highest cleavage rate, 0.053%, occurred in gastric juice simulant after 4 hours of incubation. The in vitro digestion model developed by RIVM, on the other hand, showed the cleavage of NAAX to 2,4-DMA in all three digestion simulants, but at lower rates. The maximum hydrolysis rate was 0.033%. Both models thus show very low cleavage of NAAX to 2,4-DMA; the slightly different results are probably due to the slightly different compositions and pH values of the simulants in the two in vitro digestion models.
NDPA: The cleavage of arylacetamides such as NDPA can be catalysed in vivo by the enzyme arylacetamide deacetylase (AADAC) (Fukami et al.short foret alii (lat. "and others") 2015; Yoshida et al.short foret alii (lat. "and others") 2018) . The reaction has been well studied, for example, for the drugs indiplon, flutamide and phenacetin, which are structurally very similar to NDPA (Kobayashi et al.short foret alii (lat. "and others") 2012; Shimizu et al.short foret alii (lat. "and others") 2014; Watanabe et al.short foret alii (lat. "and others") 2009) . Since in silico data generated using the OECD Toolbox Version 4.6 also indicate cleavage of the amide bond of NDPA, the BfRshort forGerman Federal Institute for Risk Assessment assumed in its original risk assessment that 2,4-dimethylaniline is released during the metabolism of NDPA. In addition, the LUA Saxony had detected 2,4-dimethylaniline in two samples in addition to NDPA (see sections 2 and 3.1.1), which also indicated a possible cleavage of NDPA. In a recent study, two different in vitro digestion models were used to investigate the potential cleavage of NDPA to 2,4-DMA (Bittner et al.short foret alii (lat. "and others") 2022) . The digestion model recommended by EFSAshort forEuropean Food Safety Authority showed a time-dependent increase in 2,4-DMA after incubation with gastric juice simulant, with the highest rate being 0.21% after 4 hours of incubation. In contrast, little or no cleavage (<0.02%) was observed in saliva and intestinal fluid simulants. In line with these results, the in vitro digestion model developed by RIVM also observed the formation of 2,4-DMA mainly after incubation with gastric juice simulant (maximum: 0.021% after 4 hours), while subsequent incubation with an intestinal fluid simulant did not result in any further increase in 2,4-DMA. Arylacetamide deacetylase (AADAC) was not included in the enzyme mix, which is why there remains uncertaintyUncertaintyTo glossary as to whether NDPA could be cleaved in vivo in the liver.
3.1.2.2 Genotoxicity/carcinogenicity
With the exception of NDPA, the BfRshort forGerman Federal Institute for Risk Assessment does not have complete original data for any of the four compounds. None of the compounds has been harmonised classified in accordance with Regulation (EC) No 1272/2008 (CLP Regulation). Furthermore, no carcinogenicity study, for example in accordance with OECD Guideline 451, has been conducted for any of the compounds. Summaries of genotoxicity studies are available for NAAX and HNS. Two in vitro tests in accordance with the OECD guidelines on genotoxicity are available for NDPA. The BfRshort forGerman Federal Institute for Risk Assessment also used the OECD Toolbox Version 4.6 and the algorithms "Derek Nexus" (Version 1.1) and "Sarah Nexus" (Version 2.0) to make in silico predictions on genotoxicity and carcinogenicity and considered possibilities for a "read-across" to structurally similar compounds. The available data and the results of the predictions and read-across are presented below.
3.1.2.3 Study data
NDPA: Two in vitro studies on genotoxicity are available: a mutagenicity test in bacteria (Ames test, in accordance with OECD Guideline 471) and an in vitro micronucleus test (in accordance with OECD Guideline 487). NDPA did not cause any change in the number of revertant colonies, regardless of metabolic activity. Similarly, NDPA did not lead to an increase in the number of human lymphocytes containing micronuclei, either with or without metabolic activation. NDPA showed no genotoxic effects under the test conditions and studies were conducted in accordance with the guidelines.
Naphthol AS: Two Japanese studies on genotoxicity are available: a mutagenicity test in bacteria (Ames test) and an in vitro chromosome aberration test (JECDB 2019a) . The English-language results tables show that the substance is not to be assessed as genotoxic in vitro. It is not clear from the English-language results tables whether there has been compliance with the relevant OECD Guideline 471 and 473. No in vivo analyses of genotoxicity are available.
NAAX: The registration dossier submitted to ECHAshort forEuropean Chemicals Agency (ECHAshort forEuropean Chemicals Agency 2019g) lists a study on mutagenicity in bacteria in accordance with OECD Guideline 471 (Ames test) and an in vitro chromosome aberration test in accordance with OECD Guideline 473. The registrants made the classification that the studies were valid and conducted in accordance with the guidelines. In both studies, no genotoxic effect of NAAX was observed with or without metabolic activation. This assessment is also shared by the "Advisory Committee on Environmentally Relevant Existing Substances" of the German Chemical Society (GDCh) (BUA 1992) . However, it is not clear whether the test conditions and the enzyme mix (S9) from rat liver used are suitable for modelling the possible cleavage of 2,4-dimethylaniline from NAAX in vivo.
HNS: The registration dossier submitted to ECHAshort forEuropean Chemicals Agency (ECHAshort forEuropean Chemicals Agency 2019i) includes a study on mutagenicity in bacteria in accordance with OECD Guideline 471 (Ames test), an in vitro chromosome aberration test in accordance with OECD Guideline 473 and an in vivo chromosome aberration test in accordance with OECD Guideline 475 in hamsters after oral administration of the substance. The studies were classified by the registrants as valid and conducted in accordance with the guidelines. One limitation described is that very few cells were examined in the in vivo chromosome aberration test. The substance was assessed as non-genotoxic in the Ames test, but as genotoxic in the in vitro chromosome aberration test (without metabolic activation). This assessment was also endorsed by the OECD (OECD 2004). In vivo, HNS showed no genotoxic properties. However, the validity of the study is very limited due to the small number of cells examined. In addition, an in vivo micronucleus test in accordance with OECD Guideline 474 is described in the literature (JECDB). The study report is written in Japanese, but the results tables and a summary are available in English. According to this, HNS is to be assessed as non-genotoxic. It is not clear from the English passages whether there was compliance with the requirements of OECD Guideline 474. Therefore, the Japanese summary extracted from the study was translated. In the methodology section, the authors report that 2000 polychromatic erythrocytes were examined per animal, which complies with the requirements of OECD Guideline 474.
In silico data
Table 2 shows the predictions for genotoxicity/carcinogenicity generated using the OECD Toolbox Version 4.6 and the algorithms "Derek Nexus" (Version 6.3) and "Sarah Nexus" (Version 3.3), including the respective Rationale. It is clear that, with the exception of HNS, all compounds have the potential to bind to proteins but not to DNA due to their chemical structure. The release of aniline derivatives discussed in section 3.1.2.1 is also classified as possible by the in silico predictions. For HNS, only the prediction that the compound could lead to the formation of micronuclei or chromosomal aberrations is positive, which the prediction algorithm provides as a rationale with the presence of a quinoid structure and the possibility of addition of a nucleophile to an α,β-unsaturated carbonyl compound (Michael addition). Neither of these seems to apply, as the compound does not have a quinoid structure and Michael addition would lead to the loss of aromaticity and would therefore be energetically unfavourable. Therefore, this alert should be assessed as irrelevant. For naphthol AS, the results were vastly negative, with the exception of Sarah Nexus, which showed a positive result with low certainty. In summary, there is no strong evidence of genotoxicity for naphthol AS. For NAAX, Derek Nexus indicates a possible risk (aromatic amines), but Sarah Nexus provides a negative result. Overall, the various tools provide conflicting results, and no clear concern for genotoxicity can be derived from them. For NDPA, Derek Nexus and the OECD Toolbox show consistent structural alerts regarding possible genotoxicity due to aromatic amines. Overall, NDPA presents the highest risk compared to the other substances examined.
Table 2
Summary of the generated in silico data
| Programm / Algorithmus | Naphthol AS | HNS | NAAX | NDPA |
| Derek Nexus | ||||
| Carcinogenicity | no alert | no alert | plausible, aromatic amine or amide | plausible, aromatic amine or amide, aniline / precursor |
| Chromosome damage in vitro | no alert | no alert | ambiguous, aniline/alkyl aniline | ambiguous, aniline/alkyl aniline |
| Mutagenicity in vitro | negative | negative | plausible, aromatic amine or amide | plausible, aromatic amine or amide |
| In vivo mutagenicity | no alert | no alert | ambiguous, aromatic amine or amide | ambiguous, aromatic amine or amide |
| Sarah Nexus | ||||
| Mutagenicity in vitro | positive, 11 certainty | negative, 29 % certainty | negative, 100 % certainty | equivocal |
| OECD Toolbox | ||||
| Bindung to DNA | no | no | no | no |
| Bindung to proteines | yes, acylation | no | yes, acylation | yes, acylation |
| Carcinogenicity | no | no | no | yes, aromatic N-acylamine (genotoxicity) |
| in vitro mutagenicity (Ames) | no | no | no | yes, aromatic N-acylamine |
| in vitro/in vivo genotoxicity (micronucleus/chromosome aberration) | yes, H-acceptor | yes, H-acceptor | yes, H-acceptor | yes, aromatic N-acylamine |
Read-across
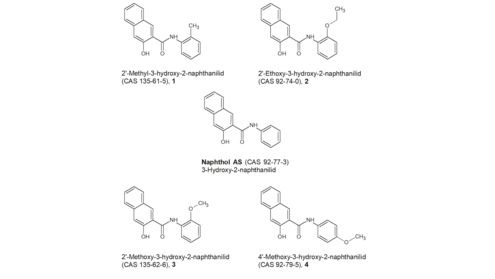
Naphthol AS: For the structurally very similar compounds 1, 2, 3 and 4 in Figure 3 , an Ames test in accordance with OECD Guideline 471 is specified in the respective ECHAshort forEuropean Chemicals Agency registration dossier (ECHAshort forEuropean Chemicals Agency 2019b; ECHAshort forEuropean Chemicals Agency 2019c; ECHAshort forEuropean Chemicals Agency 2019e; ECHAshort forEuropean Chemicals Agency 2019j). For compound 2, an in vivo micronucleus test in accordance with OECD Guideline 474 is also listed. As the BfRshort forGerman Federal Institute for Risk Assessment does not have access to the original data, it cannot verify whether there has been compliance with the guidelines – for example, regarding the bacterial strains used in the Ames test or the number of cells examined. The studies are considered valid by the respective registrants. In all tests, the tested substance was classified as non-genotoxic.
A read-across to naphthol AS appears possible on the basis of the available data. Naphthol AS itself can therefore be considered non-genotoxic. However, whether naphthol AS may be cleaved in vivo and release the genotoxic carcinogen aniline (see below) cannot be evaluated on the basis of the data. No genotoxicity was observed in the in vivo micronucleus test of compound 2 in rats, although the release of aniline is also possible there. However, if compound 2 is cleaved, 2-ethoxyaniline is released first, which, unlike aniline, is not classified as mutagenic or carcinogenic. Furthermore, there is no information available on whether naphthol AS and compound 2 are actually metabolised in the same way.
NAAX: For the structurally very similar compounds 1 (ECHAshort forEuropean Chemicals Agency 2019l) , 2 ( , 3 (Toxnet) and 4 (ECHAshort forEuropean Chemicals Agency 2019d) in Figure 4 various in vitro genotoxicity tests have been carried out, including an Ames test for all compounds in accordance with OECD Guideline 471. In vitro chromosome aberration tests in accordance with OECD Guideline 473 were also carried out for compounds 1 and 2, and gene mutation studies in mammalian cells in accordance with OECD Guideline 476 were carried out for compounds 2 and 4. As the BfRshort forGerman Federal Institute for Risk Assessment does not have access to the original data, it is unable to verify whether there was compliance with the guidelines for these studies.
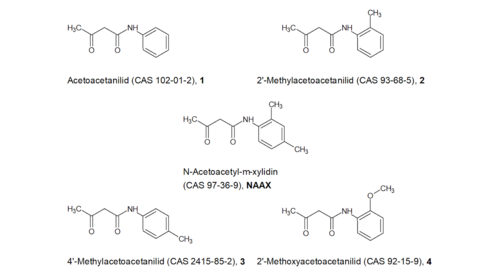
Despite isolated positive results (which, however, were always contradicted by negative results in repeat tests), the compounds are generally considered to be non-genotoxic in vitro. The results of the genotoxicity tests for NAAX (see above) are thus confirmed. However, whether NAAX may be cleaved in vivo and release the potentially genotoxic and potentially carcinogenic 2,4-dimethylaniline (see below) cannot be evaluated on the basis of the in vitro data.
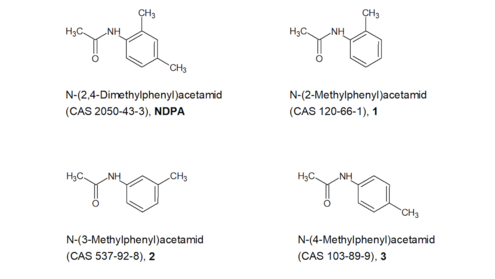
NDPA: For compound 2 in Figure 5, which is structurally very similar, a negative Ames test in accordance with OECD Guideline 471 has been published in the literature (Zeiger et al.short foret alii (lat. "and others") 1992) . The Ames test for compound 3 in Figure 5, on the other hand, was weakly positive (Zeiger et al.short foret alii (lat. "and others") 1992) . For compound 1 in Figure 5, the OECD Toolbox Version 4.1 shows a negative Ames test, which is stored in the OASIS genotoxicity database. The BfRshort forGerman Federal Institute for Risk Assessment does not have access to the original data. Compliance with the requirements of OECD Guideline 471 cannot therefore be verified. No tests on the clastogenic or aneugenic properties of the compounds are available. Whether compounds 1-3 are genotoxic cannot therefore be conclusively assessed on the basis of these data. For NDPA itself, two new genotoxicity tests are available (see above), which are compliant with the respective OECD guideline and are negative.
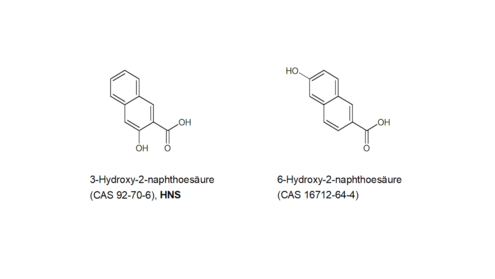
HNS: For the structurally very similar substance 6-hydroxy-2-naphthoic acid (CAS 16712-64-4, Figure 6 ), the ECHAshort forEuropean Chemicals Agency registration dossier (ECHAshort forEuropean Chemicals Agency 2019k) – analogous to HNS – specifies a positive in vitro chromosome aberration test and a negative in vivo micronucleus test. The test summary of the in vivo study states that 2000 erythrocytes per animal were examined for micronuclei. As the BfRshort forGerman Federal Institute for Risk Assessment does not have access to the original data, it is unable to verify this information.
A read-across from HNS to 6-hydroxy-2-naphthoic acid is possible due to the chemical/structural similarity. HNS is therefore not considered to be genotoxic in vivo.
3.1.2.4 Other toxicity
Naphthol AS: A Japanese screening study on SD rats for combined toxicity after repeated intake and reproductive toxicity according to OECD Guideline 422 is available (JCHECK 2019) . Doses of 0, 40, 200 and 1000 mgshort formilligram/kgshort forkilogram BW/day were administered via gavage over a period of 42 days. The English-language results tables show that, with a few exceptions for some laboratory parameters (chloride, phosphate) in the highest dose, no effects were observed. The dose at which no adverse effects were observed (NOAEL) was 1000 mgshort formilligram/kgshort forkilogram BW/day (highest dose). However, a screening study only provides an initial indication of possible effects of lifelong exposureExposureTo glossary and cannot replace a subchronic or chronic study. It is therefore not possible to derive a tolerable daily intake (TDIshort forTolerable Daily Intake) on the basis of the available data.
NAAX: A Japanese screening study on Sprague-Dawley rats for combined toxicity after repeated intake and reproductive toxicity according to OECD Guideline 422 is available (ECHAshort forEuropean Chemicals Agency 2019f; JECDB 2019b). Doses of 0, 8, 25, 80 and 250 mgshort formilligram/kgshort forkilogram body weight (BW)/day were administered via gavage over a period of 44 days. The following effects were observed: signs of methaemoglobinaemia in the two highest dose groups (reduced erythrocyte count, haemoglobin concentration and haematocrit values, and increased methaemoglobin values), as are typical of exposure to aniline derivatives (Kobayashi et al.short foret alii (lat. "and others") 2012) ; Necrosis in the spleen and increased spleen weights, deposits of haemosiderin in the liver and eosinophilic blood cells in the kidneys in the two highest dose groups. The NOAEL was 25 mgshort formilligram/kgshort forkilogram bw/day. However, a screening study only provides an initial indication of possible effects of lifelong exposure and cannot replace a subchronic or chronic study. It is therefore not possible to derive a TDIshort forTolerable Daily Intake on the basis of the available data.
NDPA: The BfRshort forGerman Federal Institute for Risk Assessment has no studies on repeated intake. As described in section 3.1.2, cleavage of the compound in the gastrointestinal tract is to be expected. Recently published data (see above) show only very slight cleavage of NDPA in digestive simulants.
HNS: The ECHAshort forEuropean Chemicals Agency registration dossier (ECHAshort forEuropean Chemicals Agency 2019i) includes a subchronic study on rats in accordance with OECD Guideline 407 and a one-generation study on rats for reproductive toxicity in accordance with OECD Guideline 415. Original data are only available for the latter in Japanese with English-language results tables (JECDB 2000) . Both studies were also subject to assessment by the OECD (OECD 2004). In the subacute study, decreased serum phosphate concentrations and increased bilirubin concentrations in the serum and urine of both sexes were observed. The females also showed slightly increased liver weights in the highest dose group and necrosis of the adrenal gland in one animal each in the highest and medium dose groups. The lowest dose of 12 mgshort formilligram/kgshort forkilogram bw/day was therefore considered to be the NOAEL. In the single-generation study, the parent generation (F0) was fed HNS via a gavage tube for a total of 98 days before mating, during pregnancy and afterwards. The study investigated effects on the offspring (F1) as well as adverse effects in the F0. The only effect observed in the offspring was a reduced mean body weight in the highest dose group (200 mgshort formilligram/kgshort forkilogram bw/day). In the F0 generation, isolated cases of liver enlargement were observed in the high-dose group. In addition, histological examination revealed hyperplasia of the mucous membrane of the forestomach. The NOAEL was 12.5 mgshort formilligram/kgshort forkilogram bw/day. A range of information, for example on individual organ weights and urine and blood parameters, as required for a subchronic study in accordance with OECD Guideline 408, is not documented in the present study. Some important information for the assessment of toxicity after repeated intake is therefore missing.
Aniline derivatives: As described in section 3.1.2.1, oral intake of naphthol AS, NAAX and NDPA may result in the release of aniline or 2,4-dimethylaniline through their cleavage. However, only a very small proportion of the substances were actually cleaved in digestive simulants.
Aniline: Aniline is classified as a mutagen (Class 2, "may cause genetic defects") and a carcinogen (Class 2, "may cause cancer") in accordance with Regulation (EC) No 1272/2008 (CLP Regulation). Although aniline was tested as non-genotoxic in the Ames test, there are positive mutagenicity tests on mammalian cells and positive in vitro micronucleus and chromosome aberration tests (ECHAshort forEuropean Chemicals Agency 2019m; EU 2004) . In vivo, the results were inconclusive. In mice, clastogenic effects were not observed or were only observed at very high, systemically toxic doses; in rats, however, these effects also occurred at significantly lower doses (ECHAshort forEuropean Chemicals Agency 2019m; EU 2004). The results of animal studies also differed to the same extent with regard to carcinogenicity. No carcinogenic effect could be detected in mice, whereas the formation of sarcomas of the spleen was observed in rats (ECHAshort forEuropean Chemicals Agency 2019m; EU 2004). In humans, aniline has been discussed as a trigger for bladder cancer since the 19th century. Modern studies have also shown an increased occurrence of bladder cancer in workers exposed to various chemicals, including aniline. However, the effect could not be clearly attributed to aniline (EU 2004).
2,4-dimethylaniline (DMA): DMA is not harmonised classified. The ECHAshort forEuropean Chemicals Agency registration dossier lists two positive in vitro chromosome aberration tests and positive Ames tests (ECHAshort forEuropean Chemicals Agency 2019h) . The BfRshort forGerman Federal Institute for Risk Assessment is not aware of any in vivo data on mutagenicity. With regard to carcinogenicity, the ECHAshort forEuropean Chemicals Agency cites an 18-month study in mice and rats. The BfRshort forGerman Federal Institute for Risk Assessment does not have the original data for any of the studies. The carcinogenicity study was not conducted under GLP and deviated in some respects from the relevant OECD Guideline 451. For example, only two dose groups were examined and only male rats were studied. Abnormalities were only observed in female mice. In the high-dose group (250 mgshort formilligram/kgshort forkilogram bw/day), the incidence of lung tumours was significantly increased compared to the control group. Since the mouse strain (CD-1) is known to spontaneously develop this form of cancer, it is questionable whether the results are transmissible to humans. Until corresponding in vivo studies are available, however, it must at least be assumed that DMA is potentially genotoxic, especially since the structurally very similar compound 2,6-dimethylaniline is harmonised as a carcinogen (class 2, "probably carcinogenic") (ECHAshort forEuropean Chemicals Agency 2019a).
3.1.2.5 Summary and derivation of acceptable intake levels
Naphthol AS: The available experimental data suggest that naphthol AS is not genotoxic in vitro. Although the in silico data indicate a certain potential for binding to DNA and proteins, studies (including in vivo studies) on chemically/structurally similar compounds have found no evidence of genotoxicity. In a screening study on toxicity after repeated intake and reproductive toxicity, no adverse effects occurred even in the highest dose group of 1000 mgshort formilligram/kgshort forkilogram bw/day. However, comprehensive data on (sub)chronic toxicity are lacking. In an earlier version of this opinion, however, the probability was high that the genotoxic carcinogen aniline would be released in the gastrointestinal tract through cleavage of the amide bond. This has now been investigated using an in vitro digestion model, and no detection of naphthol AS was possible under the test conditions used. The results were confirmed by two different digestion models, one of which corresponds to the protocol recommended by EFSAshort forEuropean Food Safety Authority. The limit of detection (LODshort forLimit of detection) for aniline in the stomach simulant is 0.9 ng/mlshort formillilitre. Using this value as the release amount for aniline, the maximum cleavage of naphthol AS was 0.023%.
Based on these data, the BfRshort forGerman Federal Institute for Risk Assessment performs an assessment of a release of naphthol AS of up to 50 µgshort formicrogram/kgshort forkilogram food as acceptable. With a cleavage of 0.023%, this would mean a maximum formation of 0.012 µgshort formicrogram aniline/kgshort forkilogram food.
We also used the margin of exposure (MOEshort forMargin of Exposure) as a tool for risk characterisation. Based on the NOAEL of 1000 mgshort formilligram/kgshort forkilogram bw/day from the above-mentioned study and the highest measured exposure value of 274 µgshort formicrogram/person/day, and applying a safety factor of 6 for extrapolation from a subacute study, a margin of exposure (MOEshort forMargin of Exposure) of 36470 was calculated. This MOEshort forMargin of Exposure value indicates a low risk to consumers, even at the highest exposure level.
NAAX: Based on the available experimental data for NAAX and structurally similar compounds, it can be concluded that NAAX is not genotoxic in vitro. However, in silico data predict a considerable genotoxic and carcinogenic potential for NAAX, mainly due to its nature as a probable aniline precursor. Corresponding in vivo metabolism studies demonstrate the release of 2,4-dimethylaniline and its subsequent binding to haemoglobin. A screening study on repeated intake toxicity and reproductive toxicity also demonstrates typical characteristics of methaemoglobinaemia, such as those that can occur after the binding of aniline derivatives to haemoglobin, in addition to effects on the liver, kidneys and spleen. In an in vitro digestion model, the highest cleavage rate of NAAX was found after a 4-hour incubation with gastric juice simulant (0.07 M HCL) and amounted to 0.053% (Bittner et al.short foret alii (lat. "and others") 2022) . Given the extremely low hydrolysis rates of the amide bonds of NAAX during simulated gastric passage, only minimal amounts of the potentially mutagenic aromatic amine 2,4-DMA are formed. Based on the available data, the BfRshort forGerman Federal Institute for Risk Assessment performs an assessment of a release of NAAX of up to 50 µgshort formicrogram/kgshort forkilogram food as acceptable. With a cleavage of 0.053%, this would mean a maximum formation of 0.016 µgshort formicrogram 2,4-DMA/kgshort forkilogram food.
NDPA: Two in vitro genotoxicity tests were performed for this compound. The Ames test, performed in accordance with OECD 471, showed no increase in revertant colonies, either with or without metabolic activation. Similarly, the in vitro micronucleus test with human lymphocytes showed no increase in the number of micronucleated cells, again with or without metabolic activation. Based on these results, NDPA was assessed as non-genotoxic. However, in silico data predict a significant genotoxic and carcinogenic potential for NDPA, mainly due to its nature as an aniline precursor, as NDPA has a high probability of being cleaved to 2,4-dimethylaniline in vivo. The reaction is catalysed by the enzyme arylacetamide deacetylase and is well described for structurally related drugs. The hydrolysis of NDPA to 2,4-dimethylaniline was investigated using an in vitro digestion model (Bittner et al.short foret alii (lat. "and others") 2022) . Under the test conditions applied, only limited hydrolysis (maximum 0.21%) was observed. Based on the available data, the BfRshort forGerman Federal Institute for Risk Assessment performs an assessment of a release of NAAX of up to 50 µgshort formicrogram/kgshort forkilogram food as acceptable. With a cleavage of 0.21%, this would mean a maximum formation of 0.078 µgshort formicrogram 2,4-DMA/kgshort forkilogram food.
HNS: Based on all experimental and in silico information on this compound and chemically/structurally similar compounds, the compound is classified as non-genotoxic. Studies on toxicity after repeated intake showed effects on the adrenal gland and hyperplasia of the forestomach mucosa. The NOAEL was 12 mgshort formilligram/kgshort forkilogram bw/day. However, a comprehensive investigation of (sub)chronic toxicity lacks a large amount of data, for example on organ weights and blood and urine values. To derive a tolerable daily intake (TDIshort forTolerable Daily Intake), a total uncertainty factor of 2000 is therefore used (a factor of 10 each for inter- and intraspecies differences, a factor of 2 for extrapolation from a subchronic to a chronic study, and a factor of 10 for uncertainties in the database). The TDIshort forTolerable Daily Intake is therefore 6 µgshort formicrogram/kgshort forkilogram bw/day. For an adult (bw = 60 kgshort forkilogram, daily intake of food that has been in contact with the respective food contact material = 1 kgshort forkilogram), this corresponds to an acceptable intake value of 360 µgshort formicrogram/day.
Acceptable migration values: In accordance with Regulation (EU) No. 10/2011, a daily consumption of 1 kgshort forkilogram of food that has been in contact with the relevant FCM is assumed for an adult. Acceptable migration values (in µgshort formicrogram/l) therefore correspond to the numerical value according to the acceptable daily intake (see Table 3 ). When using the cold water extract as a migration test, concentrations of naphthol AS, NAAX and NDPA of 50 µgshort formicrogram/l are acceptable. For HNS, a migration of 360 µgshort formicrogram/l is acceptable (see Table 3 ).
Table 3
Acceptable intake values and acceptable migration values for the water extract for naphthol AS, NAAX, NDPA and HNS
| Naphthol AS | NAAX | NDPA | HNS | |
| Acceptable intake levels for health in µg/person/day | 50 | 50 | 50 | 360 |
| Acceptable migration values in µg/l | 50 | 360 | ||
3.1.3 Exposure estimation and exposure assessment
Based on data from LUA Saxony (2018), which also determined the above-mentioned compounds (Figure 1 ) in cold water extracts from paper and cardboard matrices as part of the analysis of primary aromatic amines, a transfer of the compounds to food is to be expected upon contact with the respective FCM. In cold water extracts of FCM from paper and cardboard in accordance with DIN EN 645, naphthol AS, NAAX, NDPA and HNS were detected in the range of 2 to 368 µgshort formicrogram/l. The resulting exposure is summarised in Table 4.
While the drinking straws tested did not contain any of the organic compounds, several analytes were frequently quantified in a single sample, particularly in the baker's roll paper product group. The highest single value was determined in the water extract of a block bottom bag (HNS, 368 µgshort formicrogram/l).
In addition to these four compounds, potential cleavage products were also quantified in some cold water extracts, including the primary aromatic amines aniline and 2,4-dimethylaniline (for toxicological data, see section 3.1.2). Of note is an aniline concentration of 3 µgshort formicrogram/l in the extract of a baker's roll paper, in which the highest naphthol AS concentration of all samples was also determined (274 µgshort formicrogram/l, cf. Table 4 ). No aniline was detected in extracts with lower naphthol AS concentrations (2 to 95 µgshort formicrogram/l). This supports the assumption that naphthol AS could be a precursor to aniline. However, a statistical correlation cannot be derived from only one measurement point. A (coincidental) simultaneous impurity from both substances is also conceivable. Newly presented studies in digestive simulants showed no detectable cleavage of naphthol AS (Bittner et al.short foret alii (lat. "and others") 2022).
Table 4
Daily exposure values derived from measured concentrations of organic compounds and their potential degradation products in cold water extracts from food contact materials made of paper and cardboard (daily consumption 1 kgshort forkilogram of food).
| Model | Concentration in µg/l extract ≡ Oral exposure in µg/person/day | |||||
| Analyte | Naphthol AS | NAAX | NDPA | HNS | Aniline | 2,4-DMA |
| Total number of samples | 51 | 51 | 51 | 51 | 51 | 51 |
| Probenanzahl > Nachweisgrenze | 10 | 22 | 25 | 19 | 1 | 2 |
| Minimum value* | 2 | 2 | 2 | 7 | 3 | 3 |
| Maximum value | 274 | 55 | 22 | 368 | 3 | 3 |
| Average** | 9 | 9 | 3 | 25 | - | - |
| Median* | 7 | 13 | 6 | 24 | - | - |
- too few data points
* of samples with analyte concentrations above the limit of detection (LODshort forLimit of detection)
** Lower bound approach: Analytical content below the limit of detection (LODshort forLimit of detection) ≡ 0 µgshort formicrogram/l
2,4-dimethylaniline was detected in the cold water extracts of a napkin and a baker's roll paper sample, each containing 3 µgshort formicrogram/l. Potential starting compounds for this are NAAX and NDPA. Both compounds were quantified in the extracts containing 2,4-dimethylaniline. However, since NAAX (up to 76 µgshort formicrogram/l) and NDPA (up to 22 µgshort formicrogram/l) were found in over twenty other samples at comparable or in some cases higher concentrations, but not 2,4-dimethylaniline, an exclusive correlation between the concentrations of 2,4-dimethylaniline and NAAX or NDPA is unlikely. Newly presented studies in digestive simulants showed only very low cleavage of NAAX and NDPA (Bittner et al.short foret alii (lat. "and others") 2022).
Applying the requirement from Regulation (EU) No 10/2011, according to which the daily consumption of food, which was in contact with a respective food contact material, for an adult is 1 kgshort forkilogram, the daily exposure values listed in Table 4 are obtained.
3.1.4 Risk characterisation
As described in section 3.1.2, the BfRshort forGerman Federal Institute for Risk Assessment has derived acceptable daily intake values for naphthol AS, NAAX, NDPA and HNS (see Table 3 ). Based on current data, a daily intake of up to 50 µgshort formicrogram/person does not pose a health risk for naphthol AS, NAAX and NDPA. For HNS, a daily intake of up to 360 µgshort formicrogram/person is acceptable. As can be seen from Table 4 , NAAX and NDPA were detectable in almost half of the 51 samples tested, and naphthol AS in 10 samples. The exposure values calculated from the measured concentrations exceed the acceptable daily intake value of 50 µgshort formicrogram/person/day for naphthol AS derived by the BfRshort forGerman Federal Institute for Risk Assessment in two samples (95 µgshort formicrogram and 274 µgshort formicrogram/person/day, respectively). A health risk assessment of these release quantities is not possible based on the current data. Although the margin of exposure (MOEshort forMargin of Exposure) to the NOAEL of a subacute screening study (1000 mgshort formilligram/kgshort forkilogram body weight/day) is very large at over 200,000 (with an assumption of 60 kgshort forkilogram body weight, consumption of 1 kgshort forkilogram of food that has been in contact with the food contact material), the BfRshort forGerman Federal Institute for Risk Assessment recommends that food contact materials should not release more than 50 µgshort formicrogram naphthol AS/kgshort forkilogram food due to the limited data available.
For HNS, NAAX and NDPA, the acceptable daily intake values derived by the BfRshort forGerman Federal Institute for Risk Assessment are not exceeded, or only very slightly.
In newly submitted studies on the cleavage of naphthol AS, NAAX and NDPA in digestive simulants showed only very slight cleavage of the amides, if any, which, with a theoretical transition of 50 µgshort formicrogram amide/kgshort forkilogram food, would lead to the formation of only very small amounts of aniline or 2,4-DMA below 0.15 µgshort formicrogram/l. Some primary aromatic amines were also detected in the extracts. These were not the subject of the present assessment.
Several of the analytes were frequently found, especially in packaging paper for baked goods, so that the total content of aniline-releasing compounds may be correspondingly higher. Other sample groups, such as drinking straws, muffin tins and napkins, showed significantly lower overall releases. In each sample group, there were also samples in which no release of naphthol AS, NAAX and NDPA could be detected.
After incorporating new studies into the overall data set, the BfRshort forGerman Federal Institute for Risk Assessment considers an increased health risk from the reported transfers of HNS, naphthol AS, NAAX and NDPA into the water extract to be very unlikely.
3.2 Uncertainty analysisUncertainty analysisTo glossary
The toxicological data base for naphthol AS, NAAX and NDPA is very small. In particular, there is a lack of data on toxicokinetics and (sub)chronic toxicity. From the available in vitro data, it seems plausible or proven that the compounds mentioned release only very small amounts of aniline derivatives in the gastrointestinal tract, if any. However, uncertainties remain regarding the metabolism of the substances in the body (e.g. in the liver).
The data available for HNS is somewhat more extensive. A tolerable daily intake (TDIshort forTolerable Daily Intake) has been derived. However, existing uncertainties in the data – such as a lack of studies on the effects on individual organ weights or blood parameters – have been taken into account in its derivation.
3.3 Risk management options
For individual samples (n = 2), migration above 50 µgshort formicrogram/kgshort forkilogram food was observed for naphthol AS. The toxicological data currently available is insufficient to perform a health risk assessment for this migration. The BfRshort forGerman Federal Institute for Risk Assessment therefore recommends that no migration of naphthol AS (as well as NAAX and NDPA) above 50 µgshort formicrogram/kgshort forkilogram food should occur. Materials with a naphthol AS, NAAX or NDPA release above 50 µgshort formicrogram/l should not be used in contact with food. The release of HNS should not exceed 360 µgshort formicrogram/l. Manufacturers should test their raw materials and end products for impurities from these substances or their release and, if necessary, seek alternatives to the materials used.
4 References
BAuA (2016) Technische Regeln für Gefahrstoffe. TRGS 905. Verzeichnis krebserzeugender, keimzellmutagener oder reproduktionstoxischer Stoffe. GMBl 2016 S. 378-390 [Nr. 19], Zuletzt geändert und ergänzt: GMBl 2018 S. 259 [Nr.15] In. External Link:https://www.baua.de/DE/Angebote/Rechtstexte-und-Technische-Regeln/Regelwerk/TRGS/pdf/TRGS-905.pdf?__blob=publicationFile
Bittner N, Boon A, Delbanco EH, Walter C, Mally A (2022) Assessment of aromatic amides in printed food contact materials: analysis of potential cleavage to primary aromatic amines during simulated passage through the gastrointestinal tract. Arch Toxicol 96(5):1423-1435 doi:External Link:10.1007/s00204-022-03254-w
BUA (1992) BUA Stoffbericht 123 - Acetoacetyl-m-xylid. Beratergremium für umweltrelevante Altstoffe der Gesellschaft deutscher Chemiker
ECHAshort forEuropean Chemicals Agency (2019a) European Chemicals Agency - C&L inventory for 2,6-dimethylaniline (CAS 87-62-7). In. External Link:https://echa.europa.eu/information-on-chemicals/cl-inventory-database/-/discli/details/90282
ECHAshort forEuropean Chemicals Agency (2019b) European Chemicals Agency - Registration dossier for 2'-ethoxy-3-hydroxy-2-naphthanilide (CAS 92-74-0). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/2012/7/7/3
ECHAshort forEuropean Chemicals Agency (2019c) European Chemicals Agency - Registration dossier for 2'-methoxy-3-hydroxy-2-naphthanilide (CAS 135-62-6). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/22161/7/7/2
ECHAshort forEuropean Chemicals Agency (2019d) European Chemicals Agency - Registration dossier for 2'-Methoxyacetoacetanilide (CAS 92-15-9). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/14534/7/7/2/?documentUUID=2d2d9d34-3df6-401f-b561-60bde9c933b8
ECHAshort forEuropean Chemicals Agency (2019e) European Chemicals Agency - Registration dossier for 2'-methyl-3-hydroxy-2-naphthanilide (CAS 135-61-5). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/13267/1
ECHAshort forEuropean Chemicals Agency (2019f) European Chemicals Agency - Registration dossier for 2'-Methylacetoacetanilide (CAS 93-68-5). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/22153
ECHAshort forEuropean Chemicals Agency (2019g) European Chemicals Agency - Registration dossier for 2',4'-dimethylacetoacetanilide (CAS 97-36-9). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/14428
ECHAshort forEuropean Chemicals Agency (2019h) European Chemicals Agency - Registration dossier for 2,4-dimethylaniline (CAS 95-68-1). In. External Link:https://echa.europa.eu/de/registration-dossier/-/registered-dossier/2085/7/7/2
ECHAshort forEuropean Chemicals Agency (2019i) European Chemicals Agency - Registration dossier for 3-hydroxy-2-naphthoic acid (CAS 92-70-6). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/14175
ECHAshort forEuropean Chemicals Agency (2019j) European Chemicals Agency - Registration dossier for 4'-methoxy-3-hydroxy-2-naphthanilide (CAS 92-79-5). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/22153
ECHAshort forEuropean Chemicals Agency (2019k) European Chemicals Agency - Registration dossier for 6-Hydroxy-2-naphthoic acid (CAS 16712-64-4). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/10731/7/7/3
ECHAshort forEuropean Chemicals Agency (2019l) European Chemicals Agency - Registration dossier for Acetoacetanilide (CAS 102-01-2). In. External Link:https://echa.europa.eu/registration-dossier/-/registered-dossier/22153
ECHAshort forEuropean Chemicals Agency (2019m) European Chemicals Agency - Registration dossier for aniline (CAS 62-53-3). In. External Link:https://echa.europa.eu/de/registration-dossier/-/registered-dossier/15333/7/8
EDI (2017) Verordnung des Eidgenössischen Departement des Innern (EDI) über Materialien und Gegenstände, die dazu bestimmt sind, mit Lebensmitteln in Berührung zu kommen (Bedarfsgegenständeverordnung) - Anhang 10 (Druckfarben). In. External Link:https://www.blv.admin.ch/dam/blv/de/dokumente/lebensmittel-und-ernaehrung/rechts-und-vollzugsgrundlagen/lebensmittelrecht2017/anhang10-verordnung-materialien-kontakt-lm-gg.pdf.download.pdf/Anhang_10.pdf
EFSAshort forEuropean Food Safety Authority Panel on Food Additives F, Processing Aids, Food Micw, Aguilar F, et al.short foret alii (lat. "and others") (2008) Note for Guidance For the Preparation of an Application for the Safety Assessment of a Substance to be used in Plastic Food Contact Materials. EFSAshort forEuropean Food Safety Authority Journal 6(7):21r doi:External Link:10.2903/j.efsa.2008.21r
EU (2004) European Union Risk Assessment Report. 1st Priority List. Volume 50. Aniline. Institute for Health and Consumer Protection of the European Chemicals Bureau
Fukami T, Kariya M, Kurokawa T, Iida A, Nakajima M (2015) Comparison of substrate specificity among human arylacetamide deacetylase and carboxylesterases. European Journal of Pharmaceutical Sciences 78:47-53
doi: External Link:10.1016/j.ejps.2015.07.006
GDCh Beratergremium für umweltrelevante Altstoffe (BUA). In. External Link:https://www.gdch.de/fileadmin/downloads/Publikationen/Weitere_Publikationen/PDF/bua_infoallgemein.pdf
JCHECK (2019) Japan Chemicals Collaborative Knowledge Database - entry for 3-Hydroxy-2-naphthanilide ( 92-77-3 ).
JECDB Japan Existing Chemical Database - Test report - Exp. No. 7817 (115-182): in vivo Micronucleus test for 3-Hydroxy-2-naphthoic acid ( 92-70-6 ).
JECDB (2000) Japan Existing Chemical Database - Test report - One-geration Reproductive Toxicity Test for 3-Hydroxy-2-naphthoic acid ( 92-70-6 ).
JECDB (2019a) Japan Existing Chemical Database - entry for 3-Hydroxy-2-naphthanilide ( 92-77-3 ).
JECDB (2019b) Japan Existing Chemical Database - entry for o-Acetoacetotoluidide ( 93-68-5 ).
Kobayashi Y, Fukami T, Higuchi R, Nakajima M, Yokoi T (2012) Metabolic activation by human arylacetamide deacetylase, CYP2E1, and CYP1A2 causes phenacetin-induced methemoglobinemia. Biochemical Pharmacology 84(9):1196-1206 doi:External Link:10.1016/j.bcp.2012.08.015
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (2001) Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings1PII of original article: S0169-409X(96)00423-1. The article was originally published in Advanced Drug Delivery Reviews 23 (1997) 3–25.1. Advanced Drug Delivery Reviews 46(1):3-26, doi: External Link:10.1016/S0169-409X(00)00129-0
OECD (2004) SIDS Initial Assessment Report For SIAM 19 - 3-Hydroxy-2-naphthoic acid.
Rasper TO (2016) Primäre aromatische Amine und Präkursoren in Pigmenten, Druckfarben und bedruckten Papieren. Diplomarbeit, Technische Universität Dresden
Sabbioni ABG (1999) Biomonitoring of arylamines: haemoglobin adducts of aniline derivatives. Biomarkers 4(3):229-236 doi: External Link:http://dx.doi.org/10.1080/135475099230895
Shimizu M, Fukami T, Ito Y, et al.short foret alii (lat. "and others") (2014) Indiplon is Hydrolyzed by Arylacetamide Deacetylase in Human Liver. Drug Metabolism and Disposition doi:External Link:10.1124/dmd.113.056184
Toxnet CCRIS/Toxnet Datenbank-Eintrag für N-Acetoacetyl-p-toluidin (2415-85-2).
Watanabe A, Fukami T, Nakajima M, Takamiya M, Aoki Y, Yokoi T (2009) Human Arylacetamide Deacetylase Is a Principal Enzyme in Flutamide Hydrolysis. Drug Metabolism and Disposition 37(7):1513-1520 doi: External Link:http://dx.doi.org/10.1124/dmd.109.026567
Yoshida T, Fukami T, Kurokawa T, Gotoh S, Oda A, Nakajima M (2018) Difference in substrate specificity of carboxylesterase and arylacetamide deacetylase between dogs and humans. European Journal of Pharmaceutical Sciences 111:167-176 doi: External Link:10.1016/j.ejps.2017.09.040
Zeiger E, Anderson B, Haworth S, Lawlor T, Mortelmans K (1992) Salmonella mutagenicity tests: V. Results from the testing of 311 chemicals. Environmental and Molecular Mutagenesis 19(S21):2-141 doi: External Link:10.1002/em.2850190603